Competitive Benchmarking Tactics for the Pharmaceutical Industry in the Age of Social Media? | Q&A with Yann Guilain, Digimind
Nowhere is competitive benchmarking more important than in the global pharmaceutical industry, where the process for developing and releasing a new drug is complex, high-risk, and requires significant effort and resources.
Too often, the process of conducting a competitive analysis for pharmaceutical companies is siloed and focused primarily on internal processes. Sharing intelligence is equally important to fuel business strategies and growing market share, as well as attaining it.
Actively monitoring customers’ sentiments towards competitors’ products or even their product and communications strategies, can help pharmaceutical companies supplement traditional competitive intelligence and data sources with insights collected online around their product or service. Take drug development, for example. It is a prolonged process fraught with multiple risks, such as competitors producing similar products, as well as the possibility of drug failure.
Being aware of social trends and insights in the competitive environment will add value to the development process.
Simply put, production costs and pricing strategies aren’t the only factors – strategic innovation is key in pharmaceutical companies becoming more effective organizations. With a sound competitive intelligence (CI) and social listening strategy, pharmaceutical companies can not only gain insights into competitors’ strategies, but also an overview of relevant news, trends, and developments to fuel their marketing, sales, merger, and acquisition strategies.
Bonus: Download our guide on competitive benchmarking tactics for pharmaceutical companies.
Here, we turn to Yann Guilain, Associate and Vice President of Services at Digimind, for some insights into how pharmaceutical companies should modernize their CI methods, and more.
As an associate and customer care director, Yann brings over 25 years’ of experience in competitive intelligence, customer relationship management, social media monitoring, and market research. With 20 of those 25 years specifically with Digimind, Yann has contributed enormously to the current successes of the company and with the global expansion of becoming the powerhouse in social media and competitive intelligence it is today.
1. In what ways do you see competitive and social media intelligence benefiting the pharmaceutical industry?
The pharmaceutical industry has always benefited from competitive intelligence during the whole drug development lifecycle. Due to the highly competitive nature of the environment, pharmaceutical companies are constantly struggling to enhance research and development outlook, molecular discoveries, and lengthy and costly development and clinical trial phases. Therefore in order for these companies to mitigate these risks and the severity of the issues, they often implement competitive intelligence strategies.
Before, during, and after these phases, pharmaceutical companies often construct a competitive intelligence organization to successfully launch a product. During this process, this is when companies:
- Identify and work closely with key opinion leaders (KOLs)
- Design and strategize their key selling points, and insight management.
So naturally, CI has always been a part of the pharmaceutical industry, and is constantly evolving with new methodologies and innovations.
As for social media, this isn't some fad that is just appearing momentarily and is expected to fade away. The pharmaceutical industry, as well as other industries, need to learn how to adapt, analyze, and utilize social media if they want to have an edge over their competitors.
For instance, we’re seeing patients consistently detailing their patient/customer journey day to day, KOLs sharing their vision and thoughts online, and pharmaceutical companies frequently being mentioned on the news. Just look at the recent bankruptcy of Purdue Pharma, the makers of OxyContin, who are dealing with over 2,500 lawsuits. In this context, online monitoring is a necessity for the pharmaceutical industry.
Example of a cancer survivor documenting his treatment on social media.
2. As of now, companies within the pharmaceutical industry appear to be hesitant to embrace social media listening to its fullest capabilities, due to the extensive resources required, especially when dealing with pharmacovigilance. How do you see this changing in the future and how will it affect the industry going forward?
From a legal, ethical, and moral point of view, pharmacovigilance is just a way of life, and is crucial for a pharma company to continue to operate. Currently, the main reasons as to why drug candidates fail in phase II or phase III clinical trials is that there is insufficient efficacy results, and dire safety concerns, respectively. With a multitude of years and millions of dollars invested into these drug candidates, pharma companies simply just can’t afford to not pay attention to the adverse effects and concerns that has culminated to this point.
Pharmacovigilance did not wait for social media to exist; it has been present in the industry decades before, and will continue to be a foundation to protect patients and consumers for years to come. But with the current age of patient empowerment and the influence of social media, does it make pharmacovigilance more complex? In a sense, not really; the processes in place are still the same. Social media is just a new source in the public domain and companies have to learn how to adapt and cope with it.
Even though this is the case, there is still a key issue that causes pharmaceutical companies to be hesitant in embracing it and it involves pharmacovigilance. When dealing with pharmacovigilance, regulatory bodies of certain countries often require pharmaceutical companies to report adverse events, if ever confronted with one, and often this becomes a mandatory process. How this relates to social media is that there is just simply too much noise on the social platforms and as of now, it is difficult to scan through thousands upon thousands of mentions, and manual file adverse event reports and assess whether the complaints are deemed enough to be escalated. This one key issue remains the most vital one in order to have pharma companies completely embrace the use of social media intelligence.
3. In the age of patient empowerment, we are seeing more and more patient health information being exposed online. How do you see regulations changing because of this and its effect on the future of social media listening?
Today, patients play an increasingly active role in the pharmaceutical industry. Interests in health issues greatly increased over the years with paxtients becoming much more health conscious and desiring to be informed about existing medicines that are available, basis of approval, and how they are monitored. Due to this, patients often discuss their side effects, treatment pathways, issues, and concerns openly and actively in order to gain insight and compare their patient journeys with others in a similar situation. But with this caveat, comes with the concerns of personal health information being exposed that has the potential to negatively affect the person and their data security.
We are soon entering a period of “digital health”, and monitoring these regulatory changes will be more important than ever. New HIPAA, FDA and EMA regulatory notices are published on a regular basis and tracking these can become a key competitive advantage. Digital activities in pharmaceutical companies have to be agile and ready to adapt to the constantly changing tools and processes if needed. This is especially true when these companies are multinational corporations and crossing market lines can mean vastly different restrictions and allowances when discussing about pharmaceutical drugs.
4. Through the lens of competitive intelligence and social media listening, what innovative methods and uses do you think the industry will propel itself towards?
Slowly but surely, pharmaceutical companies get the point about positioning patients, not products at the center of their strategy. Digital media is a one-stop-shop for this new positioning. To do so, they are now investing and structuring digital activities so that they can engage and understand patients as well as they ensure compliance for a specific drug with uses approved by the FDA, EMA, or other governing regulatory bodies.
As an example, Boehringer Ingelheim developed a whole structured approach based on live chats via Twitter in order to inform, connect and build trust between consumers, pharma companies and other healthcare stakeholders.
Another example of this is that we are seeing pharmaceutical companies utilize social media listening to identify patients with astounding and impactful stories in order to highlight their patient journeys. This creates trust between the company and the consumer, and can ultimately garner another potential customer down the line.
On top of investing in social media networks, pharmaceutical companies are also delivering a true service to patients, which includes developing complementary products such as digital tools and apps which can interact, monitor, and respond to patient insights. This can come in the form of self-regulating drug delivery technologies, or health self-measurement apps for individuals. By doing so, pharmaceutical companies are able to build a relationship with consumers and provide a platform that further builds trust and loyalty.
On another front, newcomers such as Amazon are on the road to becoming a major player within the healthcare space. In 2018, they acquired PillPack, a major prescription drug delivery service that has the ability to assimilate effectively to Amazon’s current business strategy. Amazon also recently just launched Amazon care, a virtual primary care program that is exclusively for employees within the Seattle, Washington area.
Bonus: Discover competitive benchmarking tactics for pharmaceutical companies in our industry guide.
5. What are some success cases at Digimind in relation to the pharmaceutical industry?
Over the past 20 years, Digimind has helped pharmaceutical companies by providing effective strategic intelligence in order for them to gain a competitive advantage.
We offer a competitive intelligence tool that has the ability to complement key processes within the drug development cycle.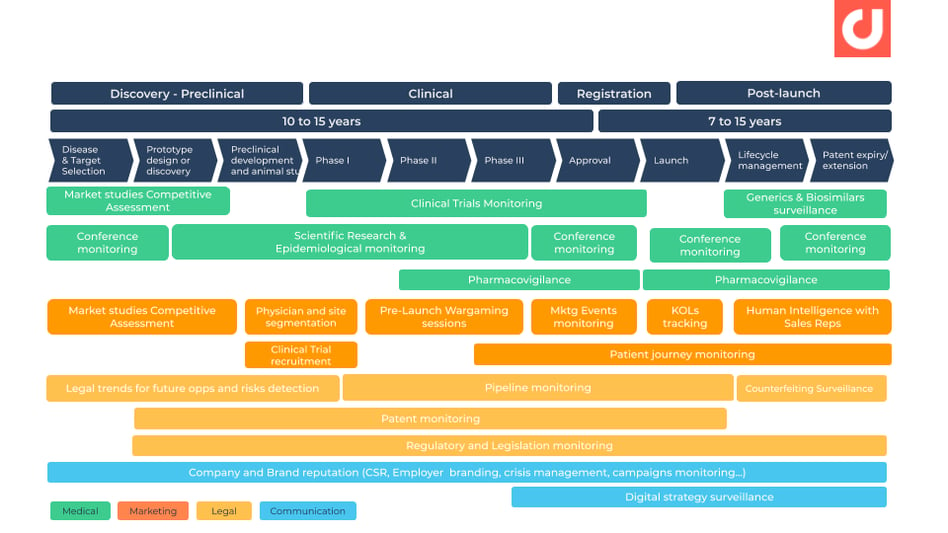 Source: Bridging Gaps in Pharmacovigilance, Digimind
Source: Bridging Gaps in Pharmacovigilance, Digimind
For example, we bring value to companies by helping them identify new molecules and biologics via monitoring of innovative biotech startups within the field.
Another success case we had appears later in the development cycle. We excel in structuring the entire drug launch phase, both from a traditional CI sense and through social media. We’re able to assist companies in obtaining analytics from their competitors in order to set up wargaming preparations.
And lastly, are cases where drugs are at the later stages of their life product cycle. Digimind Intelligence is able to monitor potential biosimilars, patent extensions, pharmacovigilance, and anticipate crisis. This is just a sample of what our pharmaceutical clients use Digimind for.
6. What are the key opportunities for social media listening in the pharmaceutical industry?
Probably the biggest opportunity for social media listening lies in its potential for real-time monitoring campaigns and tracking of activities. This is especially so as it relates to a company’s own product or its competitors'. By doing so this provides valuable insights for the company and gives them strategic leeway if any issues or concerns arise.
Second comes the monitoring of the patient’s journey. Identifying consumer expectations in terms of the level of service provided is instrumental for our clients. In this current competitive environment, pharmaceutical companies aren't solely vying for the superior product, but also for the superior “experience”’ Due to that perspective, social media is a great place to track this.
7. What are some key features from Digimind’s platform that distinguishes it from other intelligence platforms?
Digimind offers a unique combination of traditional competitive intelligence all along the drug development cycle and top social media listening capacities. Digimind’s pharmaceutical expertise allows us to guarantee full coverage of industry sources and databases, as well as top influencers on social media.
On top of that, our team of analysts take charge of the full pharmacovigilance process and comply with all our clients requirements as well as regulatory ones.
Last but not least, Digimind consistently offers new innovative resources, with a dedicated research and development team that is constantly adding new features and actively listening to input from our clients For example, a recent addition to our services is the inclusion of dark web crawling, which is mainly used to track blackmarkets and counterfeit activities.
Written by Kevin Tran
With experience in the pharmaceutical and biotechnology sector, Kevin is eager to discover new technology and methods to transcend patient healthcare. In his downtime, he enjoys traveling and playing video games.

.png?width=269&name=circle-cropped%20(1).png)